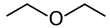Back Diëtieleter Afrikaans ثنائي إيثيل الإيثر Arabic Dietil efiri Azerbaijani دیاتیل اتر AZB Дыэтылавы эфір Byelorussian Диетилов етер Bulgarian Dietil eter BS Èter dietílic Catalan Diethylether Czech Diethylether Danish
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Ethoxyethane | |||
| Other names
Diethyl ether; Dether; Ethyl ether; Ethyl oxide; 3-Oxapentane; Diethyl oxide; Solvent ether; Sulfuric ether; Vitriolic ether; Sweet oil of vitriol
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 1696894 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.000.425 | ||
| EC Number |
| ||
| 25444 | |||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1155 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H10O | |||
| Molar mass | 74.123 g·mol−1 | ||
| Appearance | Colorless liquid | ||
| Odor | Dry, Rum-like, sweetish odor[1] | ||
| Density | 0.7134 g/cm3, liquid | ||
| Melting point | −116.3 °C (−177.3 °F; 156.8 K) | ||
| Boiling point | 34.6 °C (94.3 °F; 307.8 K)[4] | ||
| 6.05 g/(100 mL)[2] | |||
| log P | 0.98[3] | ||
| Vapor pressure | 440 mmHg at 20 °C (58.66 kPa at 20 °C)[1] | ||
| −55.1·10−6 cm3/mol | |||
Refractive index (nD)
|
1.353 (20 °C) | ||
| Viscosity | 0.224 cP (25 °C) | ||
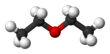
| Structure | |||
| 1.15 D (gas) | |||
| Thermochemistry | |||
Heat capacity (C)
|
172.5 J/(mol·K) | ||
Std molar
entropy (S⦵298) |
253.5 J/(mol·K) | ||
Std enthalpy of
formation (ΔfH⦵298) |
(−271.2±1.9) kJ/mol | ||
Std enthalpy of
combustion (ΔcH⦵298) |
(−2732.1±1.9) kJ/mol | ||
| Pharmacology | |||
| N01AA01 (WHO) | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Extremely flammable, harmful to skin, decomposes to explosive peroxides in air and light[1] | ||
| GHS labelling: | |||
 
| |||
| Danger | |||
| H224, H302, H336 | |||
| P210, P233, P240, P241, P242, P243, P261, P264, P270, P271, P280, P301+P312, P303+P361+P353, P304+P340, P312, P330, P370+P378, P403+P233, P403+P235, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | −45 °C (−49 °F; 228 K)[7] | ||
| 160 °C (320 °F; 433 K)[7] | |||
| Explosive limits | 1.9–48.0%[5] | ||
| Lethal dose or concentration (LD, LC): | |||
LC50 (median concentration)
|
73,000 ppm (rat, 2 hr) 6500 ppm (mouse, 1.65 hr)[6] | ||
LCLo (lowest published)
|
106,000 ppm (rabbit) 76,000 ppm (dog)[6] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 400 ppm (1200 mg/m3)[1] | ||
REL (Recommended)
|
No established REL[1] | ||
IDLH (Immediate danger)
|
1900 ppm[1] | ||
| Safety data sheet (SDS) | External MSDS | ||
| Related compounds | |||
Related ethers
|
Dimethyl ether Methoxypropane | ||
Related compounds
|
Diethyl sulfide Butanols (isomer) | ||
| Supplementary data page | |||
| Diethyl ether (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Diethyl ether, or simply ether, is an organic compound in the ether class with the formula C4H10O, (CH3CH2)2O or (C2H5)2O, sometimes abbreviated as Et2O.[a] It is a colourless, highly volatile, sweet-smelling ("ethereal odour"), extremely flammable liquid. It is commonly used as a solvent in laboratories and as a starting fluid for some engines. It was formerly used as a general anesthetic, until non-flammable drugs were developed, such as halothane. It has been used as a recreational drug to cause intoxication.
- ^ a b c d e f NIOSH Pocket Guide to Chemical Hazards. "#0277". National Institute for Occupational Safety and Health (NIOSH).
- ^ Merck Index, 10th ed., Martha Windholz, editor, Merck & Co., Inc, Rahway, NJ, 1983, p. 551
- ^ "Diethyl ether_msds".
- ^ "Diethyl ether". ChemSpider. Retrieved 19 January 2017.
- ^ Carl L. Yaws, Chemical Properties Handbook, McGraw-Hill, New York, 1999, p. 567
- ^ a b "Ethyl ether". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ a b "Ethyl Ether MSDS". J.T. Baker. Archived from the original on 2012-03-28. Retrieved 2010-06-24.
Cite error: There are <ref group=lower-alpha> tags or {{efn}} templates on this page, but the references will not show without a {{reflist|group=lower-alpha}} template or {{notelist}} template (see the help page).
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search